PNGase F
Recombinant enzyme for N-glycan removal
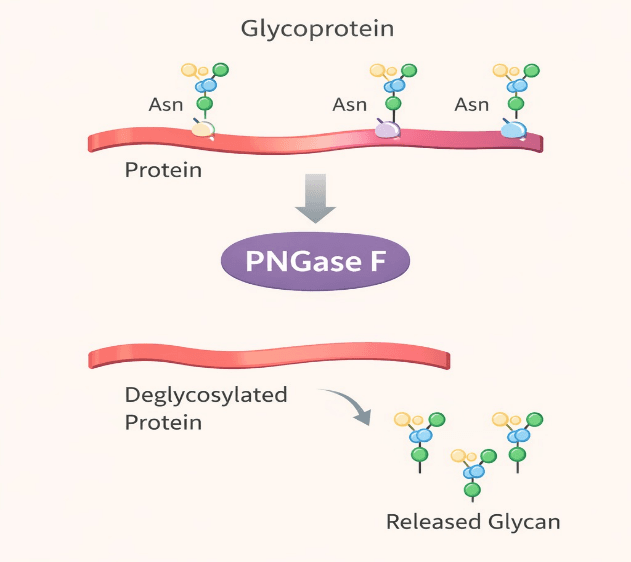
PNGase F (Peptide-N-glycosidase F) catalyzes the site-specific removal of N-linked oligosaccharides by hydrolyzing the asparagine-linked glycosylation site
Manufactured to high purity and providing excellent batch-to-batch consistency, our standard PNGase F efficiently removes high-mannose, hybrid and complex N-glycans from native or denatured proteins. It is widely used in biopharmaceutical characterization, including glycan profiling and protein mass determination, maintaining the integrity of the primary protein structure for downstream analysis.
Scientific Background
Catalyzes site-specific removal of N-linked oligosaccharides by hydrolyzing the asparagine-linked glycosylation site
Manufactured to high purity, providing excellent batch-to-batch consistency
Efficiently removes high-mannose, hybrid and complex N-glycans
Widely used in biopharmaceutical characterization and analytical biology workflows
PNGase Product Range
| Parameter | PNGase F |  |
|---|---|---|
| Primary Focus | General glycoprotein deglycosylation and structural analysis | Rapid antibody deglycosylation for high-throughput workflows |
| Reaction Time | Standard incubation tracking (2-16 hr.) | Complete N-glycan removal in ~ 10 minutes |
| Applications |
|
|
Key Features
Applications
Related Products
Explore more solutions in this category