CAS No.9014-74-8
Enterokinase
Recombinant enzyme for tag cleavage
India's Only Manufacturer
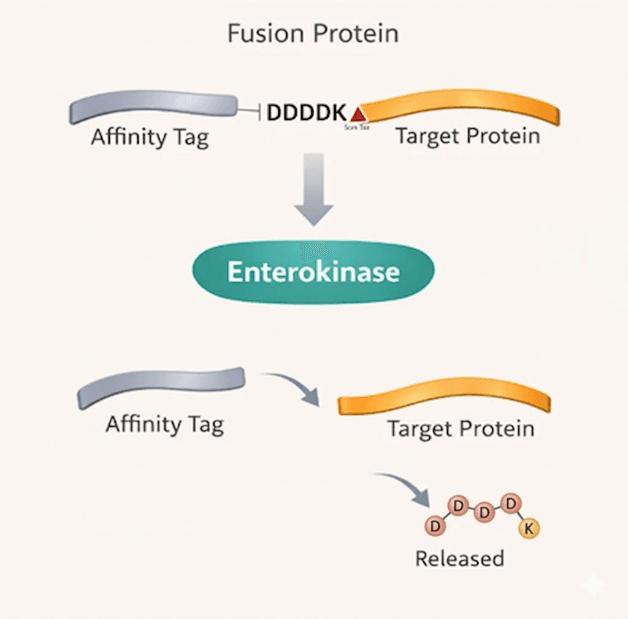
Enterokinase enables controlled cleavage of fusion tags for downstream purification and characterization workflows
A remarkably precise biochemical tool, Enterokinase perfectly recognizes and cleaves downstream of the Asp-Asp-Asp-Asp-Lys sequence marker. Functioning as a supreme sequence-specific protease, it guarantees minimal extraneous digestion, resulting in pure native target proteins. Essential for removing affinity fusion tags, its bioprocess-tolerant properties cater dynamically to stringent laboratory upscaling tasks.
Key Features
Highly Specific Cleavage at DDDDK↓ Recognition Site
Minimal Non-Specific Proteolysis at Low Concentrations
Compatible with Multiple Fusion Protein Tag Systems
Stable Activity Across a Range of Buffer Conditions
Applications
Protein PurificationRecombinant Protein WorkflowsFusion Tag Removal
Related Products
Explore more solutions in this category